The Hydrogen Economy Relies on Deloro
Introduction
In the battle against climate change, the world needs to reach net-zero emissions at least by 2050. Although there is no single path to achieve this, many of the technologies pioneered today will play a significant role. These technologies include building hydrogen value chains and decarbonization via carbon capture technologies, among others.
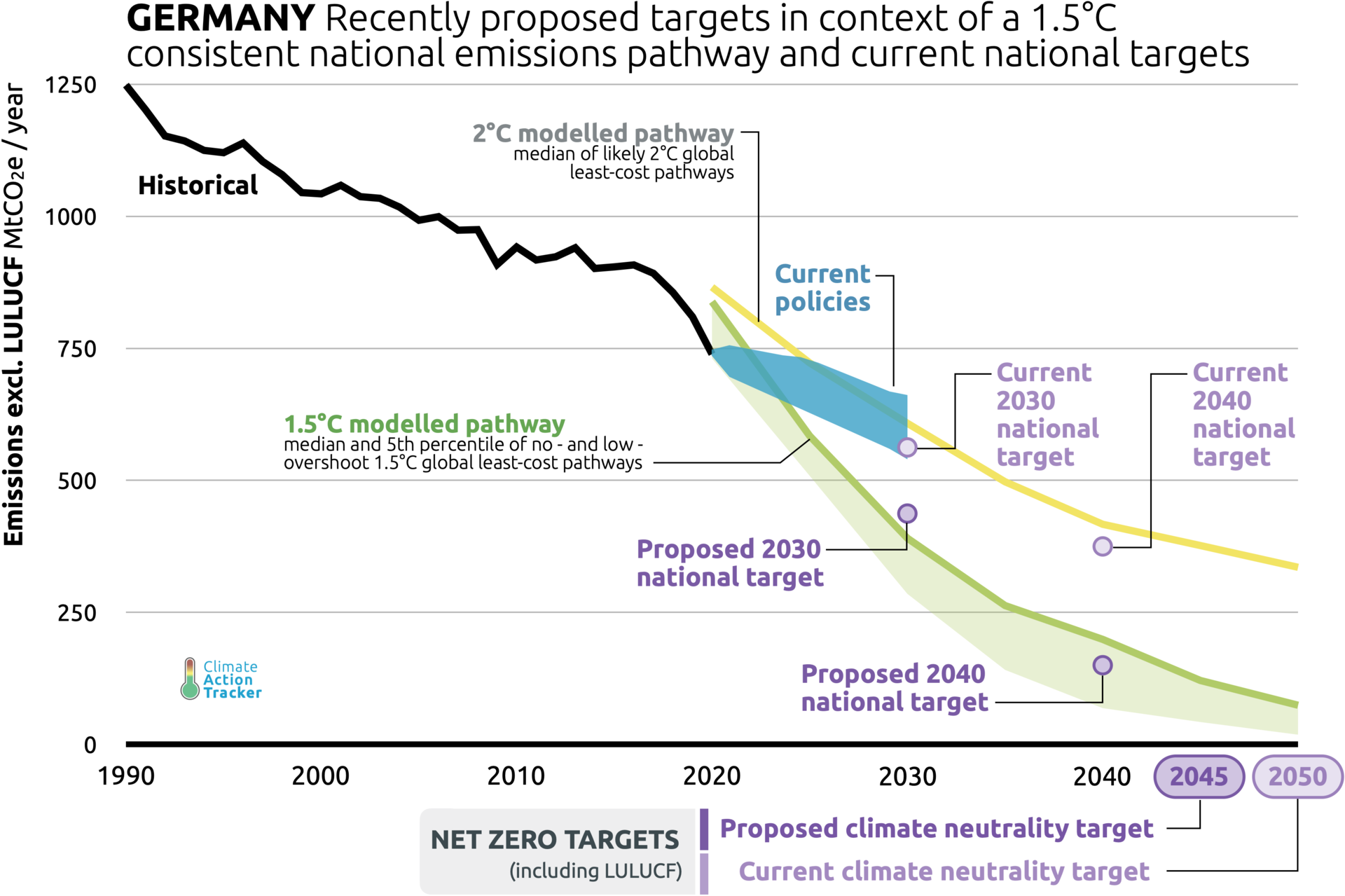
Hydrogen as a clean energy carrier holds great promise in mitigating the problems
Traditionally, Hydrogen has been used in applications such as:
- refining petroleum,
- treating metals,
- producing fertilizer,
- food processing and
- lowering the sulfur content of fuels.
In modern applications, hydrogen is increasingly being used as an energy carrier for the storage and delivery of large amounts of energy.
Modern hydrogen applications demand durable products, such as, engine components, valves/seats, nozzles, compressors and turbines, placing higher demands on their performance under extreme conditions (e.g. pressure and temperature).
These extreme conditions demand specially designed components and materials, and it is also important to ensure that metallic and non-metallic materials seals and lubricants are compatible with hydrogen.
A single material/alloy that satisfies the requirements for all processes does currently not exist. Different types of alloys are suitable for different applications, depending on the temperature and pressure requirements.
When using hydrogen gas, two major challenges arise for metallic materials (Ref. 6):
- a) severe wear due to friction – dry character of hydrogen gas –
b) tribocorrosion in a gas engine – higher amount of water vapor as combustion product - premature failure due to hydrogen embrittlement
In tribosystems, e.g. valve/seat, solid lubrication is an effective method to counteract the dry property of hydrogen as a fuel (Ref. 6). Wear resistant coatings such as WC, Cr2O3, TiN, Tribaloy™ or Deloro™ alloys, applied as functional layers, can improve friction and wear in hydrogen environments (Ref. 6).
Hydrogen embrittlement is the change in brittleness affected by the diffusion and incorporation of hydrogen into a metal (Ref. 6). Hydrogen atoms can penetrate the crystal structure of solid metal and remain embedded, increasing pressure and internal stresses without any corresponding increase in material strength. Thus, hydrogen absorption can make the metal walls brittle, which may lead to crack formation and eventually cause failure. Furthermore, tensile strength, toughness and ductility of the metal are reduced. This can result in violating the elongation-at-break requirements specified in the European Pressure Equipment Directive 2014/68 / EU.

Modern hydrogen applications demand durable products, such as engines components, valves, valve/seat, nozzles, compressors and turbines...
The use of special high-strength steels that often work in other challenging situations unfortunately does not solve this problem. On the contrary, these materials are even more vulnerable to hydrogen embrittlement. Materials that are most vulnerable include titanium and aluminum alloys.
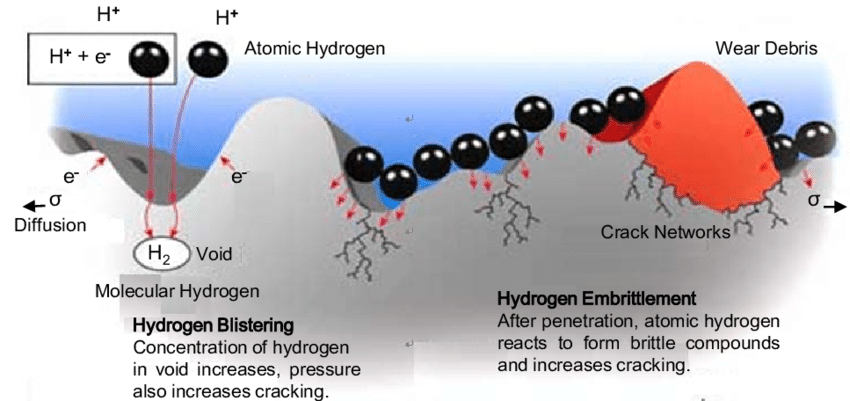
Source: https://www.researchgate.net/figure/Schematic-view-of-hydrogen-embrittlement-process-1_fig1_305926666
In the selection of suitable materials, it is also important to ensure that further processing of these materials, i.e. welding (Ref. 5), does not compromise their integrity.
Compressors
Hydrogen (H2) is the most common element in the universe and, with a molecular weight of 2.02 g/mole, is the lightest of all gases. It possesses a very high energy content per unit of weight (caloric value = ~33 kWh/kg), making it an ideal energy carrier. However, its density at atmospheric conditions is low (90 g/m3) compared to other gases, which means compression is frequently required to meet various process conditions of different applications.
To transport large amounts of hydrogen, it must be either pressurized and delivered as a compressed gas or liquefied. Compressing hydrogen often presents unique technical challenges that are not typically seen with other process gases, such as methane (CH4) or carbon dioxide (CO2). To liquefy hydrogen, a compressor is the main component of the system.
When designing a centrifugal compressor for hydrogen service, several process parameters must be considered. Because of hydrogen’s low molecular weight and high sonic velocity (see Table 1), it will have a comparatively lower pressure rise per compression stage of the compressor relative to heavier gases.
Hydrogen – 1270 m/s Oxygen – 326 m/s Carbon Dioxide – 267 m/s Methane - 446 m/s
Tab. 1. Gas - Speed of Sound (m/s)
This means that in applications with high discharge pressures, the impeller operating speed must be increased, or additional compressor stages must be added. The latter can significantly increase rotor dynamic complexity. In some instances, the maximum permissible shaft length may not provide sufficient space to incorporate the required number of stages. In such cases, the only option is to increase the impeller operating speed. However, this then requires further consideration of material strength limits.
Mechanical strength limits of the impellers are directly correlated with tip speed. The maximum allowable impeller tip speed varies depending on the specific material used and the geometry of the impeller. These material strength limitations typically are not a concern when designing compressors for service with higher molecular weight gases because the Mach numbers limits the operating speed. However, in the case of hydrogen, the mechanical strength and impeller stress levels can become limiting factors. This issue is further complicated by the potential for hydrogen embrittlement.
With the increased need and pressure to decarbonize the transportation sector, the requirements for CO2-neutral powertrains is a key factor in the diversification of future fuels. The use of hydrogen or a methanol hydrogen mix as a fuel for internal combustion engines (ICE) vehicles is a promising trend for the transport sector.
The only significant byproducts of H2 combustion are nitrogen oxides (NOx) which can be reduced to near zero when using an advanced combustion process and a relatively simple aftertreatment system. For such systems Deloro Wear Solutions GmbH also develops several solutions, such as EGR (exhaust gas recirculation) components.
Several manufacturers are working towards increasing the hydrogen-firing capabilities of their engines towards 100%. This then requires appropriate adjustment of the engine control unit and material compatibility of the engine components.
Hydrogen, ammonia or methanol – ICE (internal combustion engines)
The benefits of the Hydrogen Internal Combustion Engine (H2ICE) are the reliance on a mature industry with a huge production infrastructure, the ability to offer “flex-fuel”. These benefits will facilitate the development of the hydrogen infrastructure.
OEMs are working on a variety of future fuel engine options, such as Hydrogen, Ammonia and Methanol. They require only minor adaptations of an existing, mature technology.
Some of the challenges related to these new fuels, e.g., corrosion, stress corrosion cracking and wear from particles (size > 1 micron) may wear combustion engine parts, such as piston ring, seals, bearings, valves and nozzles.
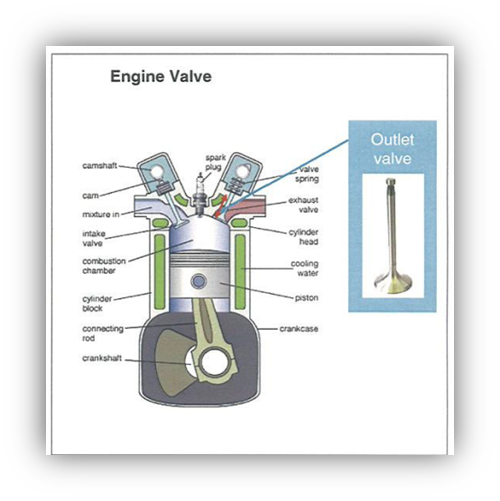
Various projects to determine the reliability of the H2ICE durability have provided a vast amount of data. Some Data was collected from an engine operated on a dynamometer for 1000 hours of continuous use. Other Data was also collected from a fleet of eight full-size pickup trucks powered with hydrogen-fueled engines (Ref. 1).The cylinder head valves and seats, as well as the injectors, were found to be particularly stressed. The intake valves showed signs of hydrogen embrittlement.
Fuel injector, nozzles, valves
The fuel injector has an injector body and an injector needle located within. Both, or at least one part thereof have a segment configured to be exposed to a combustion chamber and have a surface treated outer layer.
In general, fuel injectors are expected to last the car´s lifecycle – this cannot be assured in case of hydrogen injection.
When (liquified) hydrogen flows in large amounts at high velocity e.g. in a valve or injector, cavitation erosion occurs. There are numerous studies, simulations and tests of cavitation in liquefied hydrogen. They demonstrate (Ref 2) that in low grade steel, fast erosion occurs quickly (Ref 3). Higher cobalt content (tungsten carbide-cobalt coatings) increases the wear and cavitation resistance. (Ref 4)
Cobalt alloys show good corrosion resistance, wear resistance, and high temperature strength. These properties arise from the crystallographic nature of cobalt, the solid-solution-strengthening effects of chromium, wolfram, and molybdenum, the formation of metal carbides, and the corrosion resistance imparted by chromium.
Power generation with hydrogen turbines, a future energy system
Decarbonization involves increasing the prominence of low-carbon power generation, and with a corresponding reduction in the use of fossil fuels. This involves the use of renewable energy sources such as hydro, solar, wind and biomass energy. Enormous opportunities for bringing a fast, cost-effective decarbonization of the world can be found in the gas turbine that can run on less pure forms of hydrogen, which can be transported in any form, from liquid hydrogen to ammonia.
As gas turbines are upgraded to operate on hydrogen, it must be taken into consideration that with an increased share of hydrogen there is an increased share of steam in the system, leading to steam erosion of turbine blades. The application of high-performance alloys, corrosion-resistant alloys, will prevent corrosion or minimize damage.
Different types of alloys are suitable for different applications, depending on temperature and pressure requirements. Metallurgically, cobalt is a main alloying element in wear-resistant, corrosion-resistant, and heat-resistant alloys.
Summary
Hydrogen offers solutions for the decarbonization of many industrial sectors.
Industrial processes that run on fossil energy sources can change to green alternatives. It will be several years before H2 is available in sufficient quantities, can be transported cost-effectively, and can be used across the board in industry, transport, and power generation. Join us now in creating and implementing materials and technologies to meet future requirements…include Deloro in your H2-Journey.
Authors:
Erwin Costea - Dr.-Ing., Dipl.-Phys. / Ph.D.Mech.Eng., M.Sc.Physics
Sascha Hannappel - Dipl. Wirt.-Ing.(FH)
Jessica Karrenbrock - Dipl. Ing. (FH)
Deloro Wear Solutions GmbH
For many years, the name Deloro Wear Solutions has been associated with practical know-how and specialization in high-performance alloys that perform under difficult operating conditions such as wear, corrosion and/or high temperatures. Deloro Wear Solutions' Stellite™ cobalt alloys are already the first choice for manufacturing or coating power generation components. Feel free to contact us for more information!
Contact us
References
- Findings of Hydrogen Internal Combustion Engine Durability Final Technical/Scientific Report Project Period: 3/7/07 – 12/31/2010 Garrett P. Beauregard March 31, 2011 DE-FC26-06NT43027 Electric Transportation Engineering Corporation 430 S. 2nd Ave Phoenix, AZ 85003
- Fuel Injector Holes (Fabrication of Micro-Orifices for Fuel Injectors)
George Fenske, Nicholaos Demas, and Robert Erck: Argonne National Laboratory May 11th, 2011 - Murilo S.LamanaaAnderson G.M.PukasiewiczaSanjaySampathb a) Federal University of Technology, Paraná, Av. Monteiro Lobato Km 04, s.n., Ponta Grossa, PR 84016-210, Brazil b) Center for Thermal Spray Research, Department of Materials Science and Engineering, Stony Brook University, Stony Brook, NY 11790, USA Received 19 May 2017, Revised 5 December 2017, Accepted 11 December 2017, Available online 12 December 2017.
- Corrosion- and Hydrogen-Resistance of Heat-Resistant Blade Nickel-Cobalt Alloys, A. I. Balyts’kyi, Yu. H. Kvasnyts’ka, L. M. Ivas’kevich & H. P. Myal’nitsa, Materials Science volume 54, pages230–239 (2018)
- WELDING TECHNOLOGY, Hydrogen Embrittlement And Cracking Explained, WORKSHOP INSIDER, DECEMBER 17, 2020
- CIMAC Guideline: H2 in Stationary Gas Engines for Power Generation, 2021-10 (1st edition), 4.3 Materials

